

A battery’s positive end (cathode) and negative end (anode) are two vital components that largely define how well it can perform. In particular, researchers have focused on improving the efficacy and cost-effectiveness of cathode design, since they can account for a significant amount of the costs for producing lithium batteries.
Lithium-manganese-rich oxides are promising cathode candidates, because they don’t use cobalt – which is expensive, scarce, and fraught with issues around non-ethical mining operations. However, Jahn-Teller (CJT) distortions of Manganese ions pose a major drawback to their use. Counteracting this instability has been a major roadblock until researchers at the at the Advanced Institute for Materials Research (WPI-AIMR) at Tohoku University demonstrated a paradigm that suppresses the root cause of CJT distortions. The resulting cathode is long-lasting, with near-perfect cycling stability.
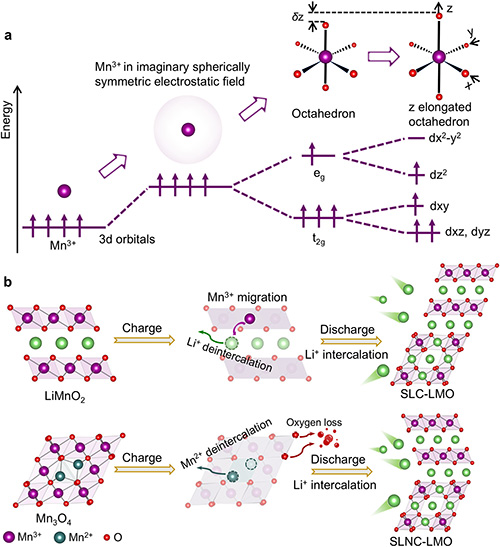
Orbital origin of Mn3+ instability and formation of collinear and noncollinear interfaces. (a) Schematic illustration of Mn3+ orbital splitting as it transitions from a free ion state to an octahedral crystal field. (b) Electrochemical formation pathways for the collinear (SLC-LMO) and noncollinear (SLNC-LMO) heterostructures. ©Hanghui Liu et al.
The scientific significance of this research lies in its innovative use of “interfacial orbital engineering” to solve structural challenges that have plagued manganese-based batteries for decades. Rather than relying on macroscopic coatings or doping, the researchers addressed the root cause at the atomic and electronic levels, utilizing “orbital geometric frustration” at noncollinear interfaces to effectively neutralize the cooperative Jahn-Teller distortions that typically lead to material collapse. This design philosophy, rooted in electronic orbital topology, not only achieved near-perfect cycling stability (zero degradation after 500 cycles) but also established a bridge between electrochemistry and solid-state physics, providing a new universal paradigm for developing high-performance, distortion-resistant energy materials.

Li+ storage behavior and cycle performance. Charge-discharge profiles of the (a) monoclinic LiMnO2 and (b) spinel Mn3O4 precursors. (c) Direct comparison of the cycle performance for the derived SLC-LMO and SLNC-LMO cathodes. ©Hanghui Liu et al.
This breakthrough heralds the arrival of cheaper, more durable, and eco-friendly battery technology in everyday life. Current batteries for electric vehicles and smartphones rely heavily on problematic cobalt; in contrast, manganese is abundant, low-cost, and environmentally benign.
This advancement means we can look forward to more affordable electric vehicles with reliable driving ranges, without the constant worry of significant battery degradation over time. More importantly, stable and inexpensive manganese-based batteries can support large-scale storage for wind and solar energy, accelerating the global transition to clean energy, reducing carbon emissions, and ensuring long-term ecological security for our living environment.
“Given the cost advantages of manganese, Mn-based oxides also represent the most commercially promising cathode materials for sodium-ion batteries,” says Distinguished Professor Hao Li (WPI-AIMR), explaining another potential direction for this innovative research in the future.
The findings were published in the Journal of the American Chemical Society on February 11, 2026.
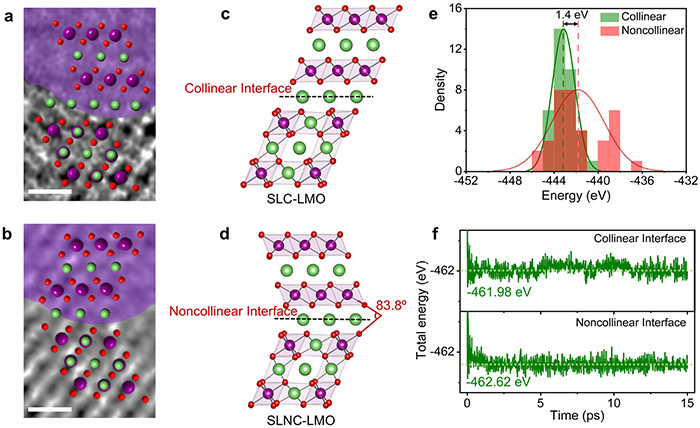
Atomic-scale interfacial structure and energetic stability of collinear and noncollinear interfaces. (a, b) ABF-STEM images of SLC-LMO and SLNC-LMO samples. Scale bar, 0.5 nm. (c, d) Corresponding atomic models of the SLC-LMO and SLNC-LMO structures. (e) Histogram showing the 0 K total energy distributions over magnetic configurations for SLC-LMO (collinear, green) and SLNC-LMO (noncollinear, red) structures. (f) Total energies as a function of MD time for SLC-LMO and SLNC-LMO structures at 300 K. ©Hanghui Liu et al.
| タイトル: | Interface-Mediated Jahn-Teller Effect in a Structure-Reinforced LiMnO2 Cathode |
|---|---|
| 著者: | Hanghui Liu, Tao Shen, Xiaohui Zhu, Mei Yang, Yushuai Yao, Eric Jianfeng Cheng, Yue Zhao, Hao Li, Shuang Li, Lin Gu, Hui Xia |
| 掲載誌: | Journal of the American Chemical Society |
| DOI: | 10.1021/jacs.5c20036 |
東北大学材料科学高等研究所(WPI-AIMR)
教授 Hao Li(研究者プロフィール)
| E-mail: | li.hao.b8@tohoku.ac.jp |
|---|---|
| Webstie: | Hao Li Laboratory |
東北大学材料科学高等研究所(WPI-AIMR) 広報戦略室
| Tel: | 022-217-6146 |
|---|---|
| E-mail: | aimr-outreach@grp.tohoku.ac.jp |