

12/26/2016
© 2016 Ali Khademhosseini
By employing a gold film that contains multiple levels of pores, AIMR researchers have produced a sensitive detector capable of monitoring low levels of reactive chemicals that are released from muscles in the body. This detector is promising for advancing our understanding of the metabolism and functionality of skeletal muscle tissue.
Muscles attached to bones generate small amounts of reactive chemicals known as superoxide anions. Normal muscular activity generates low levels of superoxide anions, but high levels can be problematic since the species is quite toxic. “High levels of superoxide anions may trigger malfunctioning, aging and damage of muscle tissue,” notes Ali Khademhosseini of the AIMR at Tohoku University.
Now, Khademhosseini and his co-workers have developed a highly sensitive detector that can detect very low levels of superoxide anions1.
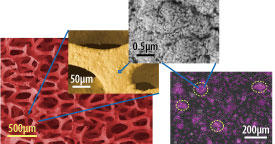
The genius of their detector lies in the fact that it uses a gold film that is porous on both macro- and nanoscales. Nanoporous gold is riddled with tiny pores and consequently has a high surface area. But, as Khademhosseini notes, “Although nanoporous gold has a large surface-to-volume ratio, only a small region of the film near the surface is exposed to the surrounding medium.”
To overcome this problem, the researchers imparted the film with a high porosity on a macroscale by fabricating nanoporous gold films on templates of nickel foam, which contained macroscale pores (see image). This greatly increased the proportion of the film that is exposed to the medium. Since nickel is toxic to the body, the team chemically stripped the template away.
The scientists then demonstrated the effectiveness of the detector by coating it with an enzyme and then using it to measure the superoxide levels generated by muscle cells from mice. The biosensor realized a sensitivity that is two to three orders of magnitude higher than previous devices.
This high sensitivity makes the detector suitable for non-invasive measurements. “Since the physiological concentrations of superoxides are remarkably low, even sensitive electrochemical biosensors must be placed close to the region of interest, making them impractical for safe, in vivo operation,” notes Khademhosseini. “Our sensor, on the other hand, can detect much lower levels of superoxides and thus can be placed farther from the site of injury.”
The researchers intend to use their biosensor to investigate the rates at which superoxide anions are released when skeletal muscle tissue is subjected to electrical stimuli. They also anticipate that the multiple-porosity strategy used in the study will be applicable to a broad range of biosensing applications.
This research highlight has been approved by the authors of the original article and all information and data contained within has been provided by said authors.