

Ammonia isn’t just for cleaning supplies and plant fertilizers – it can also serve as a precursor to clean hydrogen energy. The decomposition of ammonia (NH3) is a promising carbon-free pathway that makes hydrogen, which can be used as a fuel that doesn’t emit harmful fumes like fossil fuels do. Recent research at Tohoku University is tackling the main issue that makes ammonia decomposition challenging by creating a non-noble metal catalyst that speeds up the reaction to a practical rate.
An inexpensive nickel (Ni) catalyst doped with yttrium (Y) was created to solve these issues. The combined structure (called Ni1Ce1-xYxOα) is formulated in such a way that it can generate many, stable surface oxygen vacancies, which are vital for controlling the reaction for ammonia decomposition. Additionally, the new catalyst design allowed the research team to precisely adjust the electronic environment of Ni active sites (sites on the catalyst that other molecules can bind to in order to trigger certain chemical reactions). As a result, the catalyst greatly improved the performance of the ammonia decomposition reaction.

The structure and elements distribution. (a) TEM, (b) HRTEM, (c) STEM and elements mapping: (d) Ni and (e) overlay of fresh Ni1Ce0.5Y0.5Oα; (f) TEM, (g) HRTEM, (h) STEM and elements mapping: (i) Ni, (j) Ce, (k) Y, and (l) overlay of post-catalysis Ni1Ce0.5Y0.5Oα. (m) The schematic of the structure formation in Ni1Ce0.5Y0.5Oα after H2 pretreatment (Dark blue atoms denote Ni, red atoms denote O, yellow atoms denote Ce, and light blue atoms denote Y). ©Zhixian Bao et al.
Non-noble metal catalysts like Ni often suffer from insufficient intrinsic activity and high energy barriers for N2 desorption. However, the research team was able to create a high−performance, non−noble metal catalyst by carefully incorporating yttrium as a dual-function promoter. This study unveils how Y-doping represents a unique strategy to produce stable and effective catalysts to aid with ammonia decomposition.
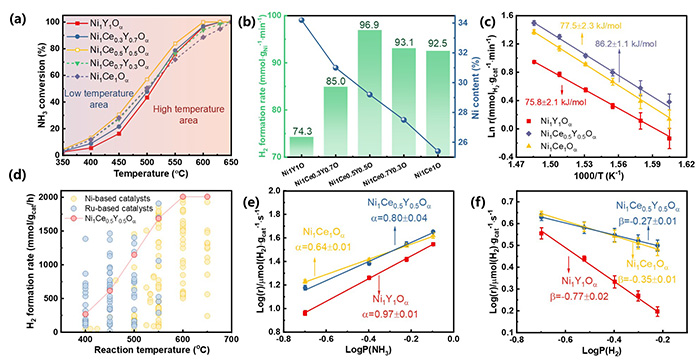
Temperature-dependent activities of Ni1Ce1-xYxOα (GHSV=30000 mL·gcat-1·h-1, reduction temperature=400 °C). (b) The H2 formation rate of Ni1Ce1-xYxOα at 500 °C based on the weight of Ni on the catalysts. (c) The apparent activation energy of Ni1Ce1-xYxOα (x=0, 0.5, 1); (d) Comparative analyses of H2 formation rate across catalysts reported in literature at the different temperatures, and the H2 formation rate of Ni1Ce0.5Y0.5Oα at the different temperatures positioned closer to the top plot. All the data are also available in the DigCat database: https://www.digcat.org/ . Dependence of the H2 formation rate on the partial pressure of (e) NH3 and (f) H2. ©Zhixian Bao et al.
. Dependence of the H2 formation rate on the partial pressure of (e) NH3 and (f) H2. ©Zhixian Bao et al.
“This study provides a practical pathway toward more sustainable, affordable hydrogen energy systems,” says Associate Professor Yizhou Zhang. “The findings support the broader transition to clean energy, contributing to reduced carbon emissions and the future deployment of hydrogen-based vehicles and power generation.”
The findings were published in Journal of Catalysis on January 30, 2026.
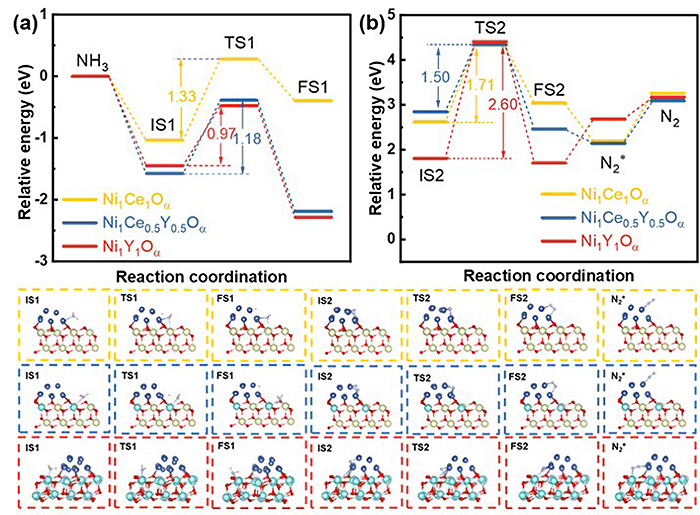
Potential energy diagrams for (a) the first N-H bond dissociation and (b) N-N coupling of adsorbed N atoms (N* + N*) to form *N-N* in NH3 decomposition. The optimized structural models for each step of Ni1Ce1Oalpha; are shown in yellow boxes. Those for Ni1Ce0.5Y0.5Oα are shown in blue boxes, and those for Ni1Y1Oα are shown in red boxes. ©Zhixian Bao et al.
| Title: | Y-Induced Oxygen Vacancy Engineering and Local Electronic Reconstruction for Enhanced Ammonia Decomposition over Ni1Ce1-xYxOα |
|---|---|
| Authors: | Zhixian Bao, Huibin Liu, Yizhou Zhang, Zhiheng Wang, Hao Li, Haoquan Hu |
| Journal: | Journal of Catalysis |
| DOI: | 10.1016/j.jcat.2026.116718 |
Yizhou Zhang (Profile of Dr. Zhang)
Advanced Institute for Materials Research (WPI-AIMR), Tohoku University
| E-mail: | zhang.yizhou.e4@tohoku.ac.jp |
|---|
Hao Li (Profile of Dr. Li)
Advanced Institute for Materials Research (WPI-AIMR), Tohoku University
| E-mail: | li.hao.b8@tohoku.ac.jp |
|---|---|
| Webstie: | Hao Li Laboratory |