

12/21/2020
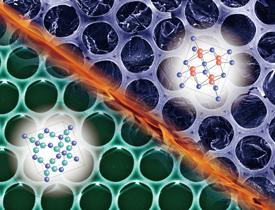
Adapted, with permission, from Ref. 1. Copyright (2020) American Chemical Society.
An ultralight material developed by AIMR researchers that can absorb waste heat and convert it directly into electricity could significantly enhance the energy efficiencies of vehicles and power plants1.
When one side of a thermoelectric material is heated, the resulting temperature gradient generates a voltage across the material. In principle, these materials should be ideal for recapturing the large amounts of energy lost as waste heat by many machines and devices. “The exhaust heat emitted from engines of automobiles and turbines of power plants is huge, and as much as 60% of it is lost to the environment,” says Hiroshi Yabu of the AIMR at Tohoku University.
However, although many thermoelectric materials are known, few operate with high efficiency in the 125–625 degrees Celsius temperature range that corresponds to the flow of exhaust heat from vehicles, gas turbines and other industrial processes.
Magnesium silicide is an exception. “Magnesium silicide is one of the few thermoelectric materials composed of Earth-abundant elements and that exhibits an optimum temperature near 425 degrees Celsius, which almost perfectly matches the temperature of industrial waste heat,” says Yabu.
A good thermoelectric material must have a high electrical conductivity, but it should also have a low thermal conductivity to ensure that its cold side stays cold and the temperature gradient is maintained.
Since magnesium silicide’s thermal conductivity is too high for practical use, Yabu and his colleagues have been developing ways to slow heat transfer through the material. “The introduction of a porous structure is an effective approach to reduce the thermal conductivity, but conventional techniques to fabricate porous magnesium silicide also reduce its electrical conductivity due to defects forming,” he says.
Yabu’s team has now come up with a method to produce magnesium silicide with a honeycomb-like structure, introducing pores into the material but not defects. The researchers had previously developed a way to make a polymer honeycomb structure — in which the pores in the polymer were templated by water droplets — and to coat this structure with silica. They have now shown that silica can be converted into magnesium silicide honeycomb by treating silica-coated honeycomb with magnesium vapor at temperatures above 725 degrees Celsius (see image).
The resulting material was so light that it floated on water — an advantage for weight-critical applications such as heat harvesting on vehicles. Critically, its thermal conductivity was 11% lower than that of bulk magnesium silicide, while its electrical conductivity did not change. “This result means that magnesium silicide honeycomb is a near-ideal thermoelectric material — it is ultralight, is made from abundant materials, and has a high thermoelectric conversion efficiency,” Yabu says.
This research highlight has been approved by the authors of the original article and all information and data contained within has been provided by said authors.