

09/26/2016
Reproduced from Ref. 1 with permission of The Royal Society of Chemistry.
By applying a new mathematical approach, AIMR researchers have gained insights into the nanostructures that mixtures of two plastics form under various conditions1.
Most commercial plastics, or polymers, consist of long chains of one type of molecule. But just as different metals can be combined to produce alloys that have superior properties to their constituent metals, so polymer properties can be tuned by mixing different polymers.
Polymers made up of two types of polymer molecules are known as diblock polymers. The two chains can arrange themselves into a wide range of different configurations as a result of the attractive and repulsive forces acting between them. Even more shapes can be generated by confining diblock polymers to different volumes, such as a sphere or cylinder. This ability to modify the shapes, and hence the properties, of diblock polymers is generating much interest because it holds promise for making tiny chemical reactors and nanoparticles for drug delivery, among other possible applications.
“Understanding the phase behaviors of diblock copolymers in confined spaces is one of the major problems in soft-matter physics,” says Hiroshi Yabu of the AIMR at Tohoku University. “Unique structures with separated microphases emerge in three-dimensionally confined spaces. While such ‘frustrated phases’ have been investigated experimentally and theoretically, little is known about the energetics of such systems.”
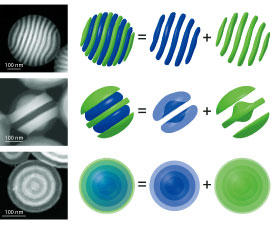
Now, Yabu and co-workers, by building on a previous mathematical analysis2, have used a set of coupled equations to numerically explore the morphologies and phases of diblock copolymers confined within spheres. They synthesized nanoparticles containing mixtures of the polymers polystyrene and polyisoprene (the polymer of natural rubber). Depending on the conditions used, the researchers obtained a variety of configurations, including some that resembled tennis balls, onion layers or hamburgers. When they compared transmission electron micrographs of diblock copolymers with predictions based on their numerical model, they found that the results were strikingly similar (see image).
“This model, which shows the relationship between the free energies and morphologies of diblock copolymers confined in small particles, has great predictive power and consistency with experimental results,” says Yabu. “It provides not only explanations of phenomena, but also reliable guidelines for designing new experiments. In particular, it could be used to suggest how to make new complex morphologies experimentally and be useful for finding new functional materials for applications such as drug delivery.
The researchers intend to use their model to explore other mixtures of polymers as well as the effect of factors such as the copolymerization ratio and molecular weight.
This research highlight has been approved by the authors of the original article and all information and data contained within has been provided by said authors.