

06/24/2013
© 2013 Mingwei Chen
The development of cheap and efficient fuel cells — devices that convert chemical energy into electricity — is a key component of the push toward cleaner energy sources. Instead of burning fuel to produce heat, fuel cells extract energy as electrons, which can then be fed into an electrical circuit. In the most basic example, the proton exchange membrane fuel cell (PEMFC), hydrogen gas is oxidized to protons and electrons at one electrode. These electrons then pass to the other electrode, via an electrical circuit, where they reduce oxygen. Lastly, the resulting oxide anions combine with the protons, which have moved through a membrane, to form water — completing the same conversion as occurs when hydrogen is burnt as a fuel.
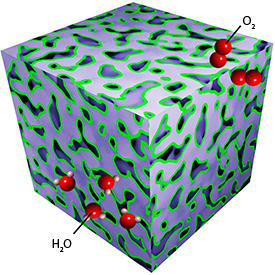
Although commercially available fuel cell systems use platinum absorbed onto carbon to catalyze the oxygen reduction reaction (ORR), the rate of the reaction is still low, meaning that a large amount of expensive and scarce platinum is required to achieve an efficient reaction. Now, Mingwei Chen and Xingbo Ge from the AIMR at Tohoku University and co-workers have created a cheap and active ORR platinum–copper catalyst1. Their system consists of a nanoporous platinum–copper alloy core covered by a thin skin of pure platinum (see image).
Previous research revealed that the precise ratio of platinum to copper in the core of these nanoparticulate catalysts is particularly important for their function. “Controlling the composition of bimetallic particles at the nanoscale is extremely difficult,” says Chen. “Our electrochemical dealloying can selectively dissolve the copper from an alloy and is much easier to control.” The dealloying process itself is governed by the voltage that is applied, allowing the final composition of the catalyst to be tuned and, in turn, the optimum catalyst to be identified.
Chen and his co-workers showed that while the catalytic activity of their material can be attributed to the thin layer of pure platinum at its surface, this property can be honed by changing the composition of the catalyst’s alloy core. This is due to the way in which the atoms in the thin layer of platinum bind to the core. Correspondingly, by adjusting the platinum–copper ratio of the core, the team was able to produce a nanoporous material that outperforms commercial platinum on carbon catalysts.
“We are now using our dealloying method to prepare other bimetallic catalysts for energy-related applications,” says Chen. “In addition, we are planning industrial collaborations to promote the practical applications of these catalysts.”
Ge, X., Chen, L., Kang, J., Fujita T., Hirata, A., Zhang, W., Jiang, J. & Chen, M. A core-shell nanoporous Pt–Cu catalyst with tunable composition and high catalytic activity. Advanced Functional Materials 23, 4156-4162 (2013). | article
This research highlight has been approved by the authors of the original article and all information and data contained within has been provided by said authors.