

Reactions and cell stack of lithium–air rechargeable batteries
久保 佳実 博士
(物質・材料研究機構 ナノ材料科学環境拠点 運営総括室 室長)
2015年6月19日(金) 16:00-17:00
AIMR本館2階 セミナー室
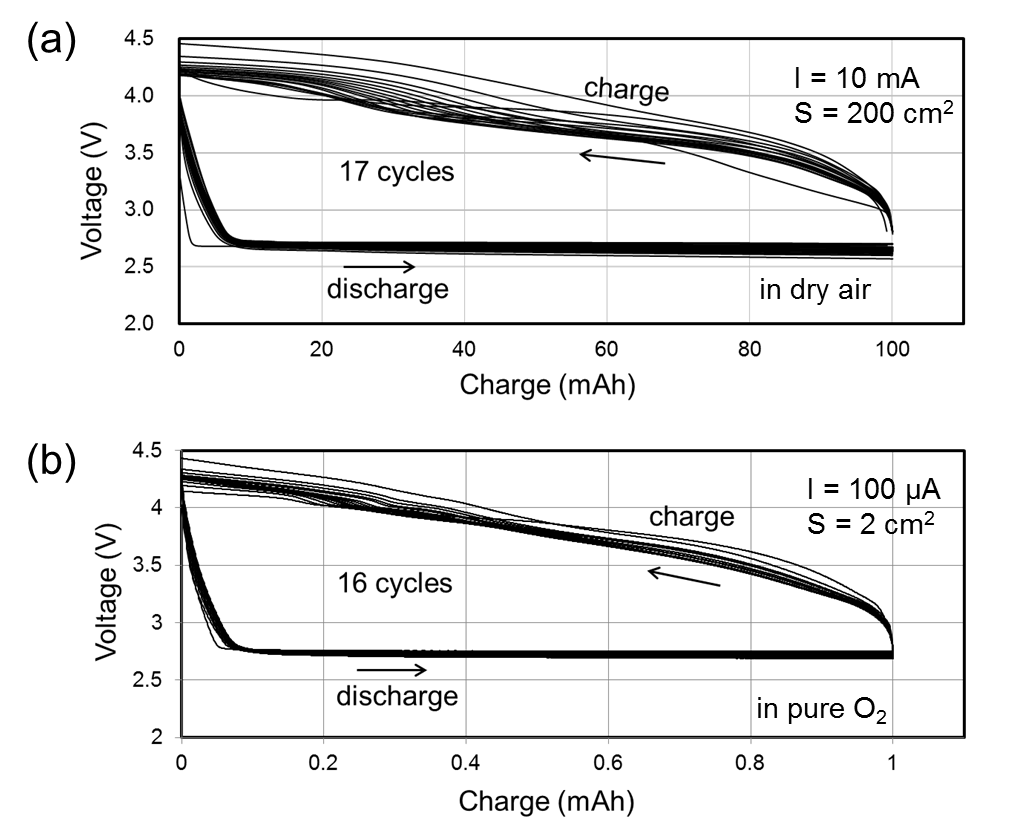
Nonaqueous lithium–air (O2) rechargeable batteries have attracted much attention because of its huge theoretical energy density, which exceeds 1000 Wh/kg. Ether-based electrolytes have been proved to be rather stable for the oxygen reduction reaction at the cathode of lithium–air batteries, showing substantially improved cyclability without apparent side reactions. However, to realize such high energy density in practical applications, multiple lithium–air cells must be densely stacked, similar to the configuration of conventional lithium-ion batteries. This stacking requires a novel design of the cell stack in which air can be supplied to the cathode. We have recently developed an “air-breathing” passive stack of nonaqueous lithium–air batteries, where air is breathed in and out through porous current collectors sandwiched between cathodes. In this seminar, I will discuss the details of cathode reactions and present the results of cell stacking.
 |
 |
|
| Figure 1. (a)(b) Schematics of the air-breathing ten-cell stack of the lithium–air battery. (c) The photo during measurement. | Figure 2. (a) Cycling performance of the ten-cell stack. (b) A parallel result for a coin cell measured at the same current density and for the same discharge capacity. |
AIMR事務部門 総務係
| TEL : | 022-217-5922 |
|---|---|
| E-MAIL : | wpi-soumu@wpi-aimr.tohoku.ac.jp |