

11/28/2016
Reproduced from Ref. 1 with permission of The Royal Society of Chemistry.
An inexpensive catalyst formed from two common metals could offer a new way to produce fuel from water, AIMR researchers have shown1.
‘Water splitting’ is an electrochemical process in which water molecules are broken apart to release hydrogen — a useful fuel. The process is a promising way to store renewably generated electricity since stored hydrogen can be used to generate electricity at times when the Sun is obscured by clouds or there is no wind. However, most electrochemical catalysts for efficiently splitting water contain rare precious metals, such as platinum, making them prohibitively expensive.
Now, by exploiting a phenomenon known as the alloying effect, Yongwen Tan from the AIMR at Tohoku University and his colleagues have developed an inexpensive alternative. Their electrocatalyst is a material called a nanoporous phosphide and contains the common metals iron and cobalt. Tan says it rivals the performances of commercial water-splitting systems based on platinum.
Alloys are materials in which one element is added to enhance the properties of another. For example, adding tin to copper creates the alloy bronze, which is stronger than copper. But alloying does not just enhance the strength of materials. “Alloying has long been known to be an effective method to create new functions of catalytic materials,” says Tan.
Tan’s team anticipated that adding iron to cobalt phosphide would alter the material’s electronic properties in such a way that its water-splitting performance would be improved. For example, according to their calculations, substituting cobalt for iron would reduce the energy of hydrogen adsorption on to the catalyst surface to the level of platinum at the optimal composition.
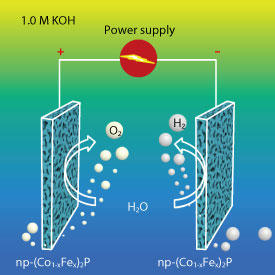
But although the calculations looked good, producing these alloys was a stumbling block. Traditional ‘wet chemistry’ methods of making alloys are unsuitable as they do not give the necessary control over the exact ratio of metals. Therefore, the researchers had to develop a novel two-step way of making them. Their process involves using an industrial technique called melt spinning to rapidly solidify the precursor alloys, controlling their composition, followed by electrochemical etching to dissolve metallic byproducts, leaving an iron−cobalt phosphide with nanoscale pores (see image) whose catalytic activity could by adjusted by fine-tuning the ratio of iron to cobalt.
The resulting electrocatalyst rivaled the performance of commercial water-splitting catalysts, but for a 20th of the price, according to Tan’s estimate. The team is now investigating the possibility of using their new fabrication technique to make a whole range of nanoporous catalysts, which could be used for many energy- and environment-related applications.
This research highlight has been approved by the authors of the original article and all information and data contained within has been provided by said authors.